 
Instead, QA should ensure quality processes are followed according to the IT quality framework defined by quality and IT system owners. 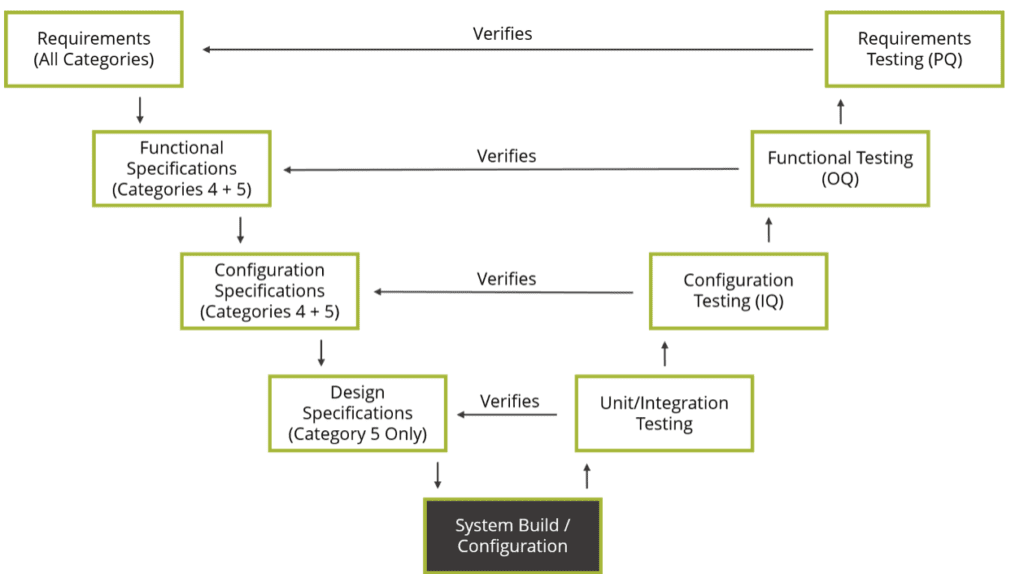
ISPE recommends that QA does not get involved with technical changes or microcode updates. Instead, the quality unit and IT system owners should establish an IT Quality function that ensures IT processes support a state of control and that IT system owners know when to inform the quality unit in the event of a change. To ensure the efficiency of infrastructure implementation and updates, this is one area where the quality unit should have a more “hands-off” role. The updated GAMP 5 also has something to say about the role of quality when it comes to IT infrastructure. Regular assessments and assurance practices, along with good IT practices, should be sufficient to handle these areas. To better monitor this service over time, it is important to define key performance indicators early.Ĭomponent-based infrastructure is generally perceived as having less risk than software applications. Lifecycle management and continual improvement will continue until decommissioning.An initial evaluation of service providers is vital for reducing risk. The recommended risk management strategy includes attention to each of the following steps: ISPE recommends a simple strategy for risk management for IT infrastructure and software tools procured by an organization. Routine assessments and good IT practices for GAMP 5 category 1 software The update GAMP 5 provides similar guidance on service provider relationships and other non-GxP tools. 
These GAMP compliance measures serve to verify the effectiveness and security of the tool or cloud service. GAMP 5’s advice in this area of non-validation is to use routine company assessment and assurance practices, along with good IT practices for non-GxP regulated systems and tools. However, that doesn’t mean life sciences companies should haven’t a practice in place for ensuring software and infrastructure security. In other words, category 1 solutions don’t need to be validated, only qualified. ISPE data integrity experts say that because GAMP 5 category 1 software does not store or create data, these tools can be managed using good IT practices and routine assessments. These non-regulated systems and tools also include cloud services, infrastructure software, and software tools that support computer system life cycle activities that do not impact patient health or product quality. GAMP 5 category 1 software includes tools used for testing or data masking. Most of these IT infrastructure and software tools are effectively off-the-shelf purchases. ISPE’s updated GAMP 5 introduces new content focusing on IT infrastructure and software tools that fall under GAMP 5 category 1, infrastructure software. In this article, we will address GAMP 5 recommendations for putting processes in place for implementing and deploying digital solutions in a GxP-regulated environment. The second edition of the GAMP 5 Good Practice Guide: A Risk-Based Approach to Compliant GxP Computerized systems, published earlier this year, offers fresh insight into the use of software tools and the importance of IT infrastructure as support and service in compliance with GxP Standards. It’s up to life science companies to strike a balance between security and efficiency to ensure an adequate state of control is maintained. However, bad practices have the potential to open a breach of vulnerable information. That’s because an investment and GAMP 5 category 1 software tools and IT infrastructure requires some level of reliance upon outside vendors that aren’t GxP-regulated.Ĭorrect application of software tools and IT infrastructure can strengthen an organization, ensuring a robust, reliable, and secure system. While the value of digital transformation is clear, implementing it while staying compliant with GxP standards is challenging. Today’s digitalization advancements are helping companies optimize and centralize their production activities, driving improvements in process quality and product safety. More life science companies than ever are coming to rely upon digital solutions. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
